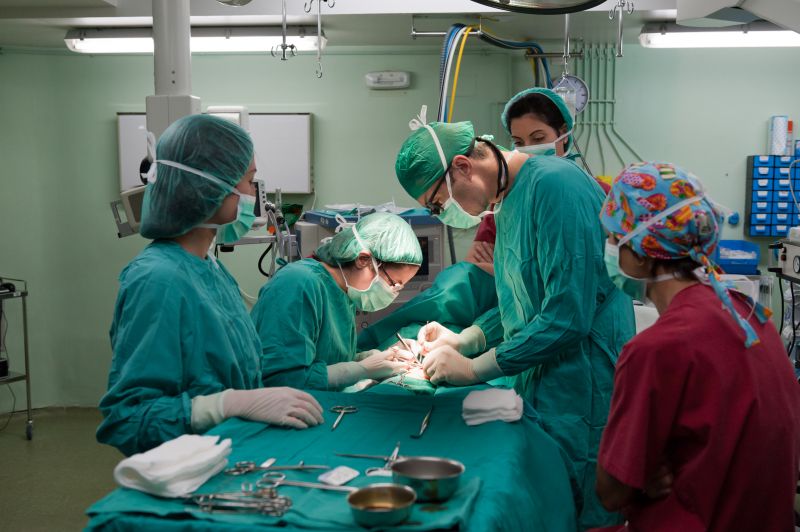
 After almost a year of debate, top U.S. Food and Drug Administration (FDA) officials warned Monday that power morcellators should not be used on most women during hysterectomy procedures. This decision is expected to significantly halt a procedure that has been reported to spread undetected cancer.
After almost a year of debate, top U.S. Food and Drug Administration (FDA) officials warned Monday that power morcellators should not be used on most women during hysterectomy procedures. This decision is expected to significantly halt a procedure that has been reported to spread undetected cancer.
The FDA used their authority to call for an immediate “black box” warning for the laparoscopic power morcellators, the strictest caution the agency issues.
Deputy director for science and chief scientist at the FDA’s Center for Devices and Radiological Health, William Maisel said, “We believe that in the vast majority of women, the procedure should not be performed.”
As we have posted on our blog for many months, there has been controversy surrounding this device. Its’ popularity rose with the increase in minimally invasive and robotic surgery that allows for tiny incisions, less blood loss and quicker recovery time. However, it has recently been discovered that the morcellators, which use a fast-spinning blade to cut up tissue from fibroids or tumors, can allow some of the tiny pieces, which may be cancerous, to spread throughout the body. Some fibroids are benign while others can be a dangerous form of cancer called uterine sarcoma, and in most cases are undetected prior to surgery. This was brought to the attention of the FDA after a Boston doctor, Amy Reed, wrote an article in the December 2013 Wall Street Journal detailing that her cancer worsened following power morcellation hysterectomy.
The new warning recommends strict limitations on the device’s use. Although the agency does not regulate physicians or medical practice, the warning could raise legal exposure for manufacturers or doctors who do not heed the FDA’s advice.
There are still some gynecologists who believe that a small number of younger women may benefit from this procedure as opposed to traditional surgery. Most believe if the morcellator can be avoided, it should be since even young women could have hidden sarcomas.
As we previously reported, Brigham and Women’s Hospital in Boston has been avocating for use of bags that surround the morcellation procedure to aid in containing cancerous tissue. On Monday they said that they stalled a four-hospital, 400-patient bag study to consider the FDA’s new guidance.
If you or a loved one has been injured as a result of a power morcellator, you may be entitled to compensation for your damages. Contact one of our Gacovino Lake attorneys at 800-246-HURT (4878) to discuss your options.